A Crucial Tool for Understanding Material Behavior
Binary phase diagrams are essential tools in materials science and engineering. They depict the phase transitions and interactions between two components under varying conditions, providing crucial insights into the behavior of metals and alloys during processes like solidification, melting, and heat treatment. This article focuses on the key concepts of binary phase diagrams, including the liquidus, solidus, solvus, and tie lines, and highlights their significance in engineering materials.
What is a Binary Phase Diagram?
A binary phase diagram is a graphical representation that shows the phases present in a two-component system as a function of temperature and composition at constant pressure. These diagrams allow engineers and scientists to predict how materials will behave when subjected to changes in temperature and composition, making them invaluable in the development and optimization of alloys.
Key Features of a Binary Phase Diagram:
- X-axis: Represents the composition of the alloy, usually as the percentage of one component in the system.
- Y-axis: Represents the temperature at which the different phases exist.
- Phases: Different regions of the diagram represent different phases (solid, liquid, or a mixture of both).
- Phase Boundaries: Lines that separate different regions, indicating where phase transitions occur (e.g., from liquid to solid or solid to a different phase).
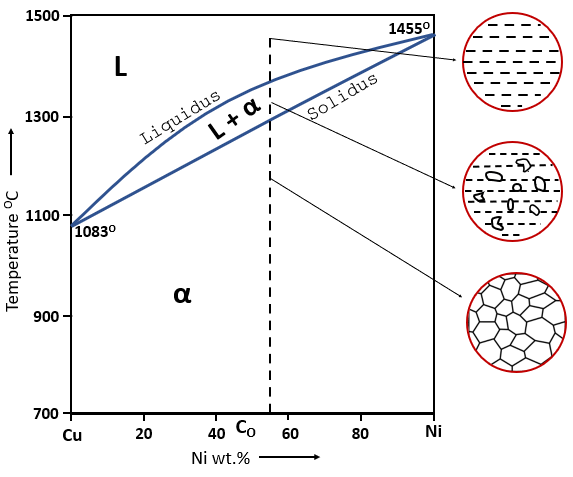
Binary Phase Diagram, Cu-Ni example (Source: Learn Metallurgy)
Key Components of a Binary Phase Diagram
Several important features of a binary phase diagram help in understanding how materials behave during phase changes. These include the liquidus, solidus, solvus, and the tie line.
1. Liquidus Line
The liquidus line is the curve in a binary phase diagram that marks the boundary above which the material is entirely in the liquid phase. Any point above this line represents a fully molten alloy. As the alloy cools and crosses this line, solidification begins.
- Definition: The liquidus line separates the fully liquid phase from the region where both solid and liquid phases coexist.
For example, in the lead-tin binary phase diagram, the liquidus line indicates the temperature at which the first solid phase begins to form as the alloy cools from the liquid state. Any temperature above this line corresponds to a fully molten alloy.
2. Solidus Line
The solidus line is the boundary below which the alloy is completely solid. Any point below this line indicates that no liquid is present, and the material exists entirely as a solid phase.
- Definition: The solidus line separates the fully solid phase from the region where both liquid and solid phases coexist.
Between the liquidus and solidus lines is the region where both liquid and solid phases are present, often referred to as the mushy zone. This region is significant in processes like casting and welding, where controlled solidification is key to achieving desired material properties.
3. Solvus Line
The solvus line indicates the boundary in a phase diagram that separates a single solid phase from a region where two different solid phases coexist. The solvus line shows the temperature and composition limits at which a second phase begins to form from a previously homogeneous solid solution.
- Definition: The solvus line represents the solubility limit of one component in another within a solid phase, distinguishing between a single-phase region and a two-phase region.
For example, in the copper-aluminum phase diagram, the solvus line shows the composition at which a second phase precipitates out of the solid solution as the temperature decreases. This is important in heat treatment processes such as precipitation hardening, where the controlled formation of a second phase strengthens the material.
4. Tie Line
The tie line (also known as an isothermal tie line) is a horizontal line drawn at a specific temperature in a binary phase diagram that connects the composition of the two coexisting phases (e.g., liquid and solid or two solid phases).
- Definition: The tie line connects the compositions of coexisting phases at equilibrium for a given temperature in a two-phase region.
For example, in a eutectic system, a tie line drawn at a temperature below the eutectic point will connect the compositions of the two solid phases (e.g., α-phase and β-phase) in equilibrium. The tie line is essential for calculating the proportions of each phase present at a specific temperature and composition, which is important in alloy design and manufacturing.
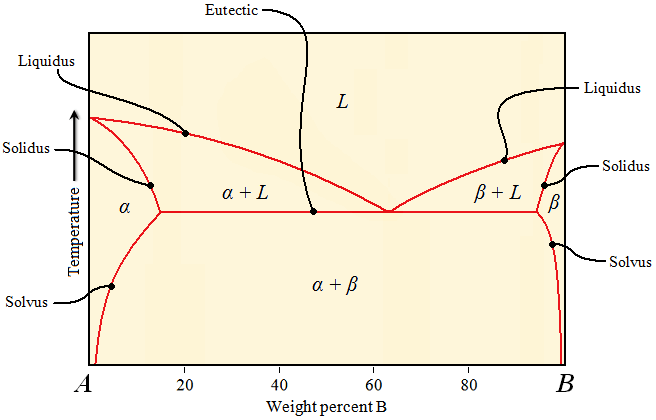
Solidus, liquidus, solvus, and eutectic lines
(Source: Engineering Archive)

Tie Line
(Source: Chemistry Stack exchange)
Importance of Binary Phase Diagrams in Engineering Materials
Binary phase diagrams are invaluable in engineering materials for several reasons. They provide critical information about how alloys behave under different thermal and compositional conditions, which is vital for optimizing material properties and processing techniques.
1. Predicting Phase Transitions
Binary phase diagrams allow engineers to predict when and how materials will transition from one phase to another. This knowledge is particularly important in processes like casting, welding, and heat treatment, where precise control over solidification and phase transformations is required to achieve desired mechanical properties.
2. Alloy Design
Engineers use binary phase diagrams to design alloys with specific properties by controlling the composition and thermal history. For example, the iron-carbon phase diagram is essential in the development of steel with varying mechanical properties. By selecting the appropriate composition and cooling rate, engineers can tailor the microstructure and, therefore, the hardness, strength, and ductility of the material.
3. Solubility and Precipitation Hardening
Binary phase diagrams provide insights into the solubility limits of one component in another, which is critical for processes like precipitation hardening. Precipitation hardening involves heating an alloy to a temperature where a second phase dissolves into the matrix, followed by cooling to precipitate out this phase and strengthen the material. The solvus line is key in determining the temperature and composition required for this process.
4. Eutectic and Peritectic Reactions
Binary phase diagrams also show reactions such as eutectic and peritectic transformations, which occur at specific compositions and temperatures. Understanding these reactions helps engineers control the microstructure of alloys, particularly in systems like aluminum-silicon and lead-tin, which are used in casting and soldering applications.